Marc Greenberg
Vernon K. Krieble Professor of Chemistry
Contact Information
- [email protected]
- Curriculum Vitae
- New Chemistry Building, 313
- 410-516-8095
- Group/Lab Website
Research Interests: Organic and Bioorganic Chemistry, Chemical Biology
Education: PhD, Yale University
Marc Greenberg is the Vernon K. Krieble Professor of Chemistry and has been a member of the Johns Hopkins University faculty since 2002. Greenberg received his training as an American Cancer Society Postdoctoral Fellow at Caltech in the laboratory of Professor Peter Dervan and earned his Ph.D. at Yale University under the guidance of Professor Jerome Berson. He has undergraduate degrees in Chemistry and Chemical Engineering from New York University and the Cooper Union School of Engineering, respectively.
Greenberg is a Fellow of the American Association for the Advancement of Science. He was the recipient of an Arthur C. Cope Scholar Award from the American Chemical Society (2016) for his research on nucleic acids, and was an Alfred P. Sloan Foundation fellow (1996-2000). Greenberg was the Founding Director of the Chemistry-Biology Interface program (2005-2013) at Johns Hopkins University.
Nucleic acids play vital roles in health and biotechnology. Our research group uses organic chemistry, biochemistry, and molecular biology to understand and exploit the reactivity and structure of nucleic acids. Our ability to chemically synthesize unnatural molecules and carry out physicochemical and biochemical studies with them enables us to address a variety of biologically important questions at the molecular level.
The investigations carried out in our group are designed to answer fundamental problems concerning nucleic acid chemistry, such as providing chemical insight into how cytotoxic antitumor agents that target DNA kill tumor cells. Practical applications give rise to tools that are useful in biotechnology. To bring these projects to fruition we synthesize novel molecules and study their behavior using a variety of physicochemical, biochemical, and biological techniques.
Recent examples include:
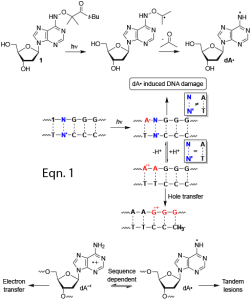
- Determining how nucleic acids are oxidatively damaged by synthesizing molecules that enable us to independently generate reactive intermediates at defined sites (eqn. 1).
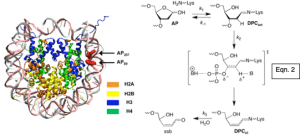
- Demonstrating that the histone proteins in nucleosome core particles catalyze DNA cleavage at damaged sites (e.g. AP, eqn. 2), and are themselves modified during the process (eqn. 3).
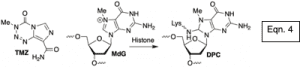
- Determine how histone proteins alter the reactivity of damaged DNA in nucleosome core particles and cellular DNA (eqn. 4).
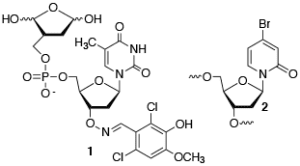
- Developing novel irreversible inhibitors of DNA repair enzymes (1).
- Developing radiosensitizing agents that enhance the effects of ionizing radiation on DNA (2).
Organic Chemistry I – 030.205
Honors Organic Chemistry II – 030.212
Advanced Mechanistic Organic Chemistry I – 030.625
Advanced Mechanistic Organic Chemistry II – 030.626
Organic Chemistry of Nucleic Acids – 030.682
Selected publications
Histone Tail Sequences Balance Their Role in Genetic Regulation and the Need to Protect DNA Against Destruction in Nucleosome Core Particles Containing Abasic Sites. Yang, K.; Greenberg, M. M. ChemBioChem 2019, 20, 78-82.
Histone Tails Decrease N7-Methyl-2'-Deoxyguanosine Depurination and Yield DNA-Protein Crosslinks in Nucleosome Core Particles and Cells. Yang, K.; Park, D.; Tretyakova, N. Y.; Greenberg, M. M. Proc. Natl. Acad. Sci. USA 2018, 115, E11212-E11220. Highlighted in Chemical & Engineering News. Highlighted in NIEHS Environmental factor, papers of the month of February.
Independent Generation of Reactive Intermediates Leads to an Alternative Mechanism for Strand Damage Induced by Hole Transfer in Poly•(dA-T) Sequences. Sun, H.; Zheng, L.; Greenberg, M. M. J. Am. Chem. Soc. 2018, 140, 11308-11316. (PMC: 6135700)
Mechanistic Insight Through Irreversible Inhibition: DNA Polymerase q Uses a Common Active Site for Polymerase and Lyase Activities. Laverty, D. J.; Mortimer, I. P.; Greenberg, M. M. J. Am. Chem. Soc. 2018, 140, 9034-9037. (PMC: 6085753)
Traceless Tandem Lesion Formation in DNA From a Nitrogen-Centered Purine Radical. Zheng, L.; Greenberg, M. M. J. Am. Chem. Soc. 2018, 140, 6400-6407. (PMC: 5966344)
DNA Damage Emanating From a Neutral Purine Radical Reveals the Sequence Dependent Convergence of the Direct and Indirect Effects of g-Radiolysis. Zheng, L.; Greenberg, M. M. J. Am. Chem. Soc. 2017, 139, 17751-17754. (PMC: 5729073)
Synergistic Effects of an Irreversible DNA Polymerase Inhibitor and DNA Damaging Agents on HeLa Cells. Paul, R.; Banerjee, S.; Greenberg, M. M. ACS Chem. Biol. 2017, 12, 1576-1583. Correction: ACS Chem. Biol. 2018, 13, 832-832. (PMC: 5492961) (Highlighted (“Spotlight”) in Chem. Res. Toxicol. 2017, 30, 1367-1368.)
The A-Rule and Deletion Formation During Abasic and Oxidized Abasic Site Bypass by DNA Polymerase q. Laverty, D. J.; Averill, A. M.; Doublié, S.; Greenberg, M. M. ACS Chem. Biol. 2017, 12, 1584-1592. (PMC: 5499511)
Rapid Histone Catalyzed DNA Lesion Excision and Accompanying Protein Modification in Nucleosomes and Nucleosome Core Particles. Weng, L.; Greenberg, M. M. J. Am. Chem. Soc. 2015, 137, 11022-11031.
Light-Triggered RNA Annealing by an RNA Chaperone. Panja, S.; Paul, R.; Greenberg, M. M.; Woodson, S. A. Angew. Chem. Int. Ed. 2015, 54, 7281-7284.
Rapid RNA Strand Scission Following C2'-Hydrogen Atom Abstraction. Paul, R.; Greenberg, M. M. J. Am. Chem. Soc. 2015, 137, 596-599. (PMC: 4313564)